gcsescience.com 25 gcsescience.com
Instrumental
Methods
Infra-red
Spectroscopy - Identifying
Compounds.
Infra-red light is absorbed in a substance by vibrating atoms.
The frequency of the
absorbed light
is the same as the frequency at which the atoms vibrate.
The frequency at which the
atoms vibrate depends on
the mass of the
particular atom and the next atom it is joined to.
For example a carbon atom joined to another carbon
atom C - C
will
vibrate at a different frequency
from
a carbon atom joined to a hydrogen
atom C - H.
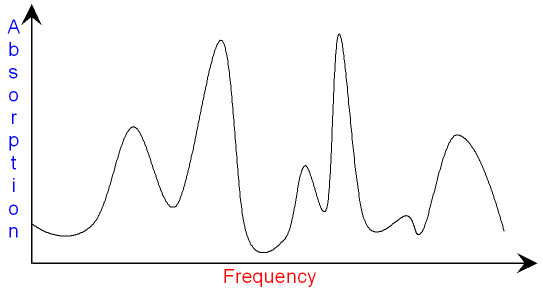
In infra-red spectroscopy all of
the different frequencies of
infra-red
light are passed through
a substance
and the instrument produces an
infra-red spectrum
showing peaks where the light has been absorbed.

The substance is identified by a
computer
which compares the shape of the
infra-red spectrum
with the spectrum of thousands
of known materials.
The area of the peak shows the concentration of the substance
(how much is present).
Infra-red spectroscopy can identify biological compounds
and may be used by the health service
and drug companies.
![]() Links
Water
Revision Questions
Links
Water
Revision Questions
![]()
gcsescience.com The Periodic Table Index Water Quiz gcsescience.com
Home GCSE Chemistry GCSE Physics
Copyright © 2015 gcsescience.com. All Rights Reserved.