gcsescience.com 22 gcsescience.com
The Transition Metals - Hydrogenation of Vegetable Oils.
What is the Hydrogenation of Vegetable Oils?
The
transition metal
nickel
is used in the
hydrogenation of
vegetable oils to make
margarines and
soft-spreads.
Hydrogenation means
adding
hydrogen to a substance.
Liquid vegetable
oils that are unsaturated will react with hydrogen
at about 60 °C in the presence of a nickel
catalyst.
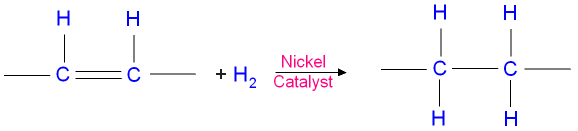
This is an example of an addition reaction where hydrogen
adds across the double
bond leaving only single
bonds.
The picture below shows hydrogenation
of a double
bond.

Hydrogenation
raises the melting point above
room temperature
and makes the liquid oil become
solid
in a process called hardening.
The solid product is
used as a margarine or spread.
![]() Links
Chemistry Quizzes
Revision Questions
Links
Chemistry Quizzes
Revision Questions
![]()
gcsescience.com The Periodic Table Index Periodic Table Quiz gcsescience.com
Home GCSE Chemistry GCSE Physics
Copyright © 2015 gcsescience.com. All Rights Reserved.