
Heating
gcsescience.com 12 gcsescience.com
Elements, Compounds and Mixtures
How do Particles Move inside a Solid, Liquid and Gas?
Solid
Liquid
Gas



Heating![]()
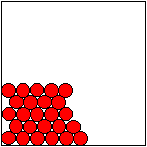
Particle Motion in a Solid.
In a solid, the particles
can vibrate
but they cannot move from one place to another.
If the solid is heated, the particles
vibrate more and more
until the force of
attraction between them is overcome.
The temperature at which this happens
is called the melting
point.
Above this temperature, the solid has become a liquid.
Particle Motion in a
Liquid.
In a liquid the force of
attraction between the particles
is weaker than it is in the
solid.
It is still strong enough that the
particles are held
close to each other but they are now free to move.
If the liquid is heated, the particles
move faster and faster
until they overcome the force of attraction
between them.
The temperature at which this happens
is called the boiling
point.
Above this temperature, the liquid has become a gas.
A liquid can also become a gas by evaporation.
What is Evaporation?
Some of the particles
in a liquid are moving slow
and some are moving fast. Some of the particles
are moving fast enough to
break through the surface
of the liquid and enter the air as a gas.
This happens
at a temperature below the liquid's
boiling
point.
For example, water will evaporate at room temperature
(25
°C)
but the boiling point
of water is 100
°C.
The faster
moving particles
are hotter
than the slower ones.
As the faster (hotter)
particles leave the liquid by
evaporation, they take heat
away from the liquid.
This is the reason that evaporation has a cooling
effect.
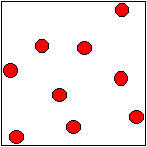
Particle Motion in a
Gas.
A gas
takes up a lot more space (occupies a greater volume)
than the boiling liquid it came from. This is called expansion.
In a gas, the particles move fast
in random directions.
There is no force of attraction
between the particles.
![]() Links
Solid
Liquid
Gas
Revision Questions
Links
Solid
Liquid
Gas
Revision Questions
![]()
gcsescience.com The Periodic Table Index Chemistry Quiz gcsescience.com
Home GCSE Chemistry GCSE Physics
Copyright © 2015 gcsescience.com. All Rights Reserved.