gcsescience.com 3 gcsescience.com
Water, Carbon and Nitrogen Cycles
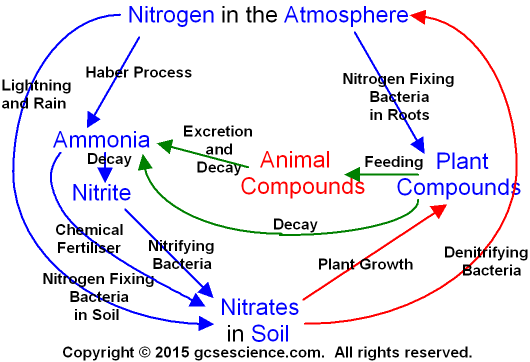
The Nitrogen Cycle.
What is the Nitrogen Cycle?
The nitrogen cycle
describes the movement of nitrogen
through the environment. Nitrogen exists in
the soil,
in plants and animals,
and in the air. Nitrogen
moves
through the processes shown in the picture
below.

How does the Nitrogen Cycle work?
Although nitrogen
gas (N2) makes up 78%
of the air, plants
can
not use it
for growth unless it is turned into nitrate
(NO3) in the soil.
Making nitrogen in the atmosphere
into nitrate in the soil is
called fixing. Nitrogen fixing occurs naturally in four ways.
Nitrogen fixing also occurs with the Haber
process - see below.
1. Nitrogen fixing
bacteria
in the soil turn nitrogen
in the soil into nitrate.
2. Nitrogen fixing
bacteria on some plant root nodules
turn nitrogen into nitrate.
Plants that have these
kinds of root nodules include peas,
beans and clover.
3. Compounds
containing ammonia occur
in
animal excretion and in dead
animals. The ammonia
turns
into nitrite and there are nitrifying
bacteria in the soil that turn
nitrite (NO2)
into nitrate (NO3).
They are called nitrifying
bacteria because they increase the amount of
nitrate in the soil.
4. Lightning can cause chemical
reactions in the atmosphere
that make nitrogen react
with oxygen producing nitrous
oxides.
Nitrous
oxides are also made from petrol engine pollution.
The nitrous
oxides can react with more
oxygen and dissolve in
rain water to make dilute nitric
acid (HNO3(aq))
(see acid
rain).
HNO3
contains nitrate and so rain
water
containing HNO3
increases the amount of nitrate in the soil.
Most of the fixing of nitrogen occurs through the
natural processes described above.
The Haber process
for
making fertilisers accounts for about 30%
of nitrogen fixing.
Nitrate in the soil is
essential for plant
growth.
There are denitrifying
bacteria
in the soil that turn
nitrate (NO3)
into nitrogen gas (N2)
and the nitrogen gas
goes back into the atmosphere. They are
called
denitrifying bacteria because
they decrease
the amount of nitrate in the soil.
Compare this with
the nitrifying
bacteria described above at number 3.
![]() Links
The Atmosphere
Revision Questions
Links
The Atmosphere
Revision Questions
![]()
gcsescience.com The Periodic Table Index Nitrogen Cycle Quiz gcsescience.com
Home GCSE Chemistry GCSE Physics
Copyright © 2015 gcsescience.com. All Rights Reserved.