CH3OH(aq) + CH3CO2H(aq)
gcsescience.com 52 gcsescience.com
The Reaction of Ethanoic Acid with Alcohols to make Esters.
Ethanoic
acid will react with
alcohols in the presence of
concentrated sulfuric
acid, to form esters.
Concentrated sulfuric acid is a catalyst for this
reaction.
methanol + ethanoic acid ![]() methyl ethanoate + water.
methyl ethanoate + water.
CH3OH(aq) + CH3CO2H(aq) ![]() CH3CO2CH3(aq) + H2O(l)
CH3CO2CH3(aq) + H2O(l)
The structure of methyl
ethanoate.
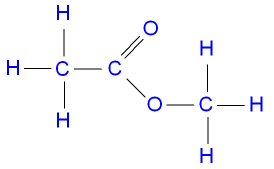
ethanol + ethanoic acid ![]() ethyl ethanoate + water.
ethyl ethanoate + water.
C2H5OH(aq) + CH3CO2H(aq) ![]() CH3CO2C2H5(aq) + H2O(l)
CH3CO2C2H5(aq) + H2O(l)
The structure of ethyl
ethanoate.
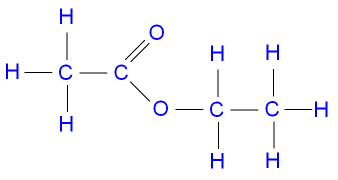
propanol + ethanoic acid ![]() propyl ethanoate + water.
propyl ethanoate + water.
C3H7OH(aq) + CH3CO2H(aq) ![]() CH3CO2C3H7(aq) + H2O(l)
CH3CO2C3H7(aq) + H2O(l)
The structure of propyl
ethanoate.
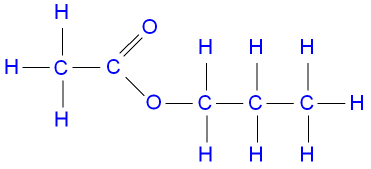
butanol + ethanoic acid ![]() butyl ethanoate + water.
butyl ethanoate + water.
C4H9OH(aq) + CH3CO2H(aq) ![]() CH3CO2C4H9(aq) + H2O(l)
CH3CO2C4H9(aq) + H2O(l)
The structure of butyl
ethanoate.
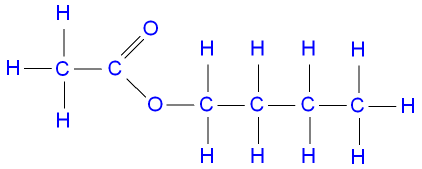
![]() Links Esters Revision Questions
Links Esters Revision Questions ![]()
gcsescience.com The Periodic Table Index Esters Quiz gcsescience.com
Home GCSE Chemistry GCSE Physics
Copyright © 2015 gcsescience.com. All Rights Reserved.