gcsescience.com 31 gcsescience.com
What is an Ammonia Molecule?
Nitrogen and hydrogen are both non-metals.
A nitrogen atom has 5
electrons in its
outer
shell.
Nitrogen is in group 5 of the periodic table.
A hydrogen atom has 1 electron in its
outer shell.
Hydrogen can only form 1 bond.
Three hydrogen atoms each share their 1
electron with
nitrogen to form three covalent bonds
and make
an ammonia molecule
(NH3).
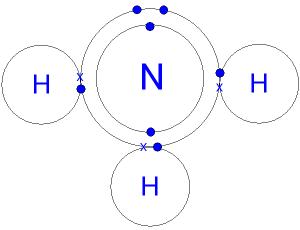
This is a picture of an ammonia molecule.

By sharing the two electrons where the shells touch
each hydrogen atom can count 2 electrons in its outer shell
and the nitrogen atom can count 8 electrons in its outer
shell.
These full outer
shells with their shared electrons
are now stable,
and the
NH3 molecule will
not
react further
with other hydrogen
or nitrogen atoms.
The molecule can also be shown
without the circles
for the shells.
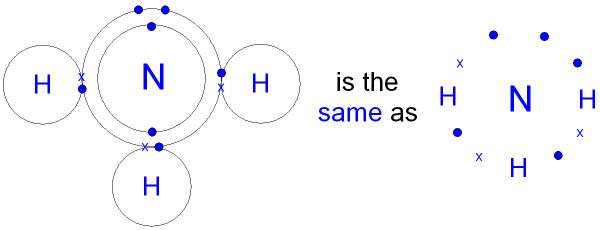
Note the 3
pairs (6 electrons)
shared between the atoms.
Each electron pair is one bond.
This
is called a single
covalent bond.
Ammonia has three single covalent bonds.
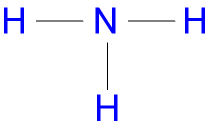
The structural formula of an ammonia molecule is written
There are no ions present (no + or - charges)
in ammonia
gas
because
the electrons are shared,
not
transferred from one atom to
another.
Ammonia does make some
hydroxide
ions
when it is dissolved
in water to form a weak
alkali.
![]() Links
Revision Quizzes
Revision Questions
Links
Revision Quizzes
Revision Questions
![]()
gcsescience.com The Periodic Table Index Covalent Bonding Quiz gcsescience.com
Home GCSE Chemistry GCSE Physics
Copyright © 2015 gcsescience.com. All Rights Reserved.