gcsescience.com 5 gcsescience.com
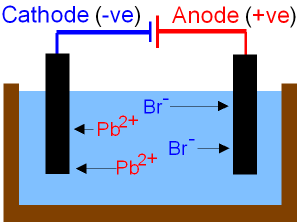
Electrolysis of Lead Bromide.
Lead
bromide must be heated until it is molten
before it will conduct electricity.
Electrolysis
separates the
molten ionic compound into its
elements.

The reactions at each electrode are called half
equations.
The half equations are written so that
the same number
of electrons
occur in each equation.
Pb2+ + 2e- ![]() Pb (lead
metal at the (-)cathode).
Pb (lead
metal at the (-)cathode).
2Br- -
2e-
![]() Br2
(bromine gas at the (+)anode).
Br2
(bromine gas at the (+)anode).
Lead
ions
gain
electrons (reduction) to form
lead atoms.
Bromide ions lose
electrons (oxidation) to form
bromine atoms.
The bromine atoms combine to form molecules of
bromine gas.
The overall reaction is
PbBr2(l)
![]() Pb(s)
+ Br2(g)
Pb(s)
+ Br2(g)
See some other examples of electrolysis.
![]() Links
Electrolysis
Revision Questions
Links
Electrolysis
Revision Questions
![]()
gcsescience.com The Periodic Table Index Metal Quiz gcsescience.com
Home GCSE Chemistry GCSE Physics
Copyright © 2015 gcsescience.com. All Rights Reserved.